Our legal team is dedicated to standing up for injured individuals and aggressively advocating for the justice and compensation they deserve.
Ozempic

Have you or a loved one developed Gastroparesis from taking Ozempic?
If you or a loved one have experienced gastrointestinal side effects after using Ozempic, which contains the active ingredient semaglutide, you may be entitled to compensation. Often, the side effect is Gastroparesis (Stomach Paralysis). This disorder slows or stops the movement of food from your stomach to your small intestine, even though there is no blockage in the stomach or intestines. Our experienced legal team is dedicated to helping individuals who have suffered from health issues related to this medication. We understand the potential challenges Ozempic poses and its association with gastrointestinal concerns, and we are here to guide you through the legal process.
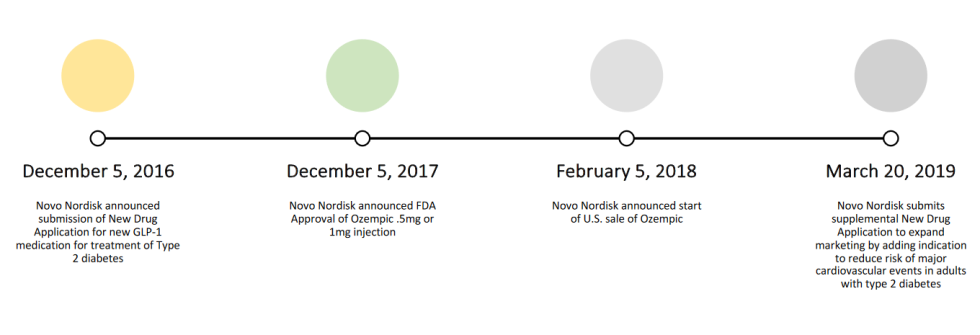
Regulatory Timeline – Ozempic
Warnings

- Vomiting and Gastroparesis – lost in the shuffle.
- Gastroparesis is not in there.
- Vomiting is in there but linked to other conditions.
- More recent labeling (2023) zeroing in on vomiting and gastroparesis.
Vomiting (OZ)
“12-2017: Product label don’t disclose the issues around the Gastroparesis”
Launch Label
Instructions
Inform patients of the potential risks and benefits of OZEMPIC and of alternative modes of therapy. Inform patients about the importance of adherence to dietary instructions, regular physical activity, periodic blood glucose monitoring and A/c testing, recognition and management of hypoglycemia and hyperglycemia, and assessment for diabetes complications. Advise patients to seek medical advice promptly during periods of stress such as fever, trauma, infection, or surgery as medication requirements may change.
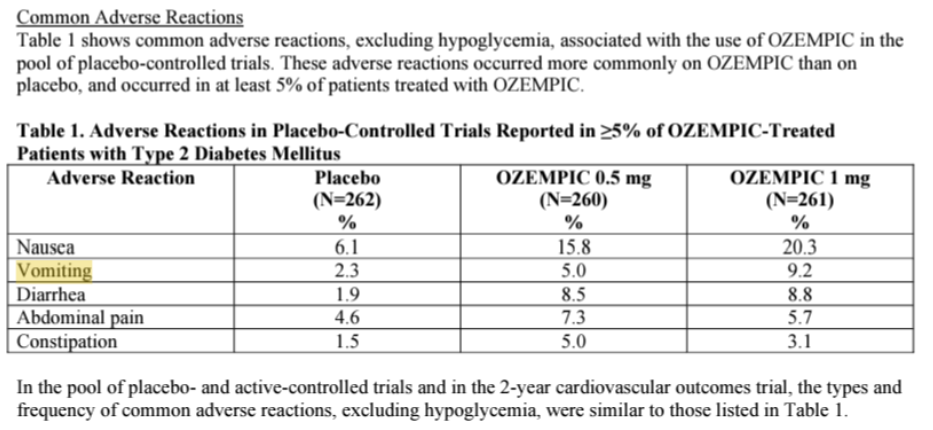
Advise patients that the most common side effects of OZEMPIC are nausea, vomiting, diarrhea, abdominal pain and constipation. Inform patients that nausea, vomiting and diarrhea are most common when first starting OZEMPIC, but decreases over time in the majority of patients.
Instruct patients to reread the Medication Guide each time the prescription is renewed.
Inform patients if a dose is missed, it should be administered as soon as possible within 5 days after the missed dose. If more than 5 days have passed, the missed dose should be skipped and the next dose should be administered on the regularly scheduled day. In each case, inform patients to resume their regular once weekly dosing schedule.
Manufactured by:
Novo Nordisk A/S
DK-2880 Bagsvaerd
Denmark
For information about OZEMPIC contact:
Novo Nordisk Inc.
800 Scudders Mill Road
Plainsboro, NJ 08536
1-888-693-6742
Date of Issue: December 2017
Version: 1
OZEMPIC and NovoFine ure registered trademarks of Novo Nordisk A/S.
PATENT INFORMATION: http://novonordisk-us.com/patients/products/product-patents.html
© 2017 Novo Nordisk
PATIENT COUNSELING INFORMATION
Patients should be advised to carefully review the FDA-approved Medication Guide and Instructions for Use. They should be informed that semaglutide has been shown to cause thyroid C-cell tumors in rodents, although the relevance to humans is unknown, and instructed to report symptoms such as a lump in the neck, hoarseness, difficulty swallowing, or shortness of breath. Patients must also be made aware of the potential risk of pancreatitis and told to discontinue Ozempic immediately and contact a physician if severe abdominal pain—possibly radiating to the back and accompanied by vomiting—occurs. Any changes in vision during treatment should be promptly reported due to the risk of diabetic retinopathy complications.
Novo Nordisk: Statistics and Facts
Novo Nordisk A/S, headquartered in Bagsvaerd, Denmark, derives a substantial portion of its revenue from diabetes and obesity-related products. In 2022, approximately 88 percent of the company’s revenue was generated from these therapeutic areas. The company reported a 26 percent increase in worldwide sales between 2021 and 2022, despite noting a decline in insulin sales during that period. Ozempic experienced significant commercial growth, with a reported 77 percent increase in sales year over year. Additionally, overall sales of obesity care products—including Ozempic, Saxenda, and Wegovy—rose by approximately 101 percent, reflecting rapid expansion within the weight management treatment market.



Contact The Michael Brady Lynch Firm
If you or someone you care about has sustained serious injuries as a result of another party’s negligence, we encourage you to reach out for a private and confidential case review.
The Michael Brady Lynch Firm
127 West Fairbanks Ave. #528
Winter Park, Florida 32789
📞 888-585-5970
Get Trusted Legal Insights
Join our mailing list to receive helpful legal resources, firm updates, and important announcements. We’re here to keep you informed every step of the way.